What if you could stop rheumatoid arthritis before it even begins? For decades, this seemed impossible. But a groundbreaking 2026 study has just shattered that assumption—proving that a simple 12-month treatment can delay disease onset by up to four years.
This isn’t just incremental progress. This is a complete paradigm shift in how we approach autoimmune diseases.
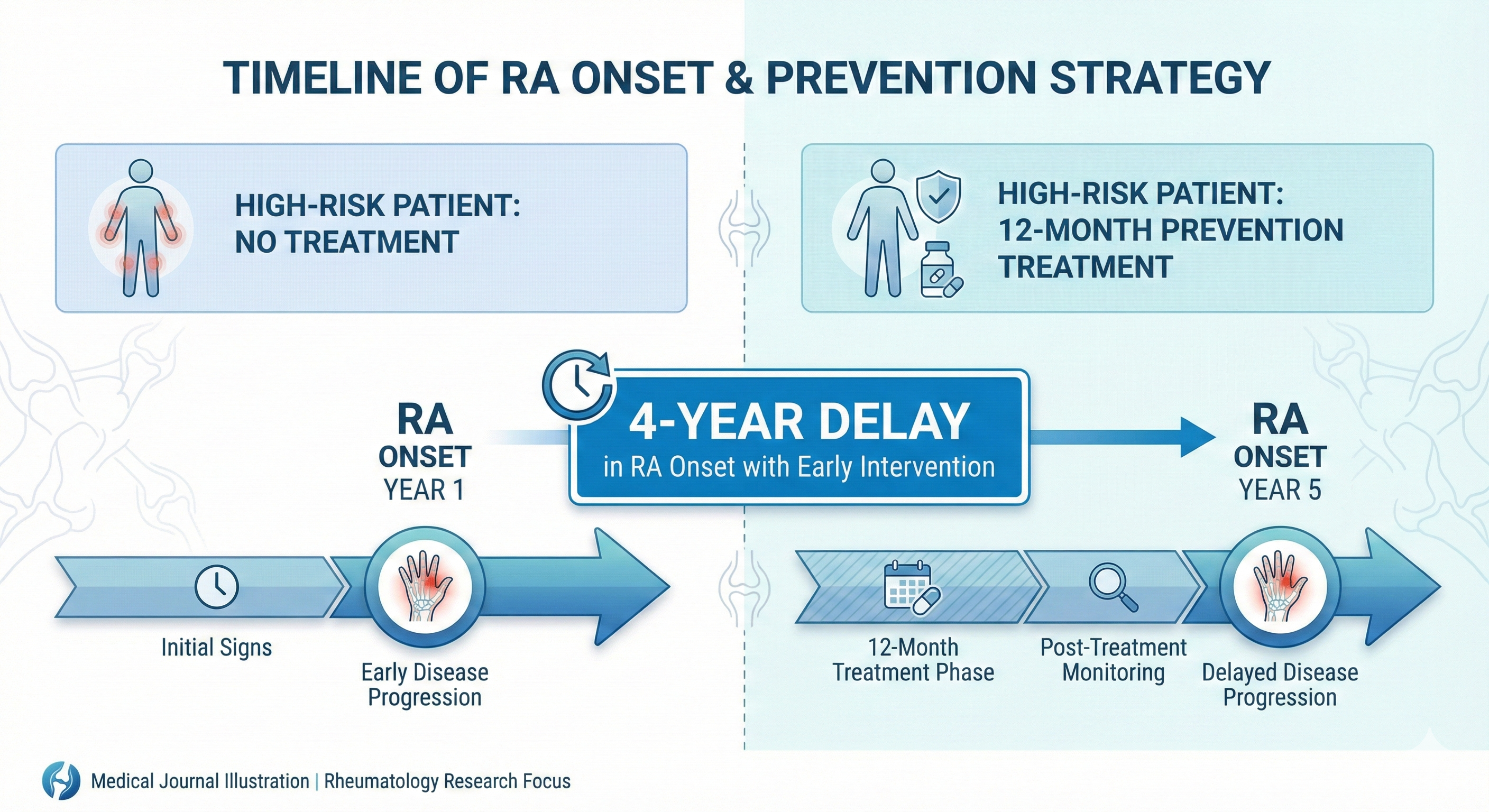
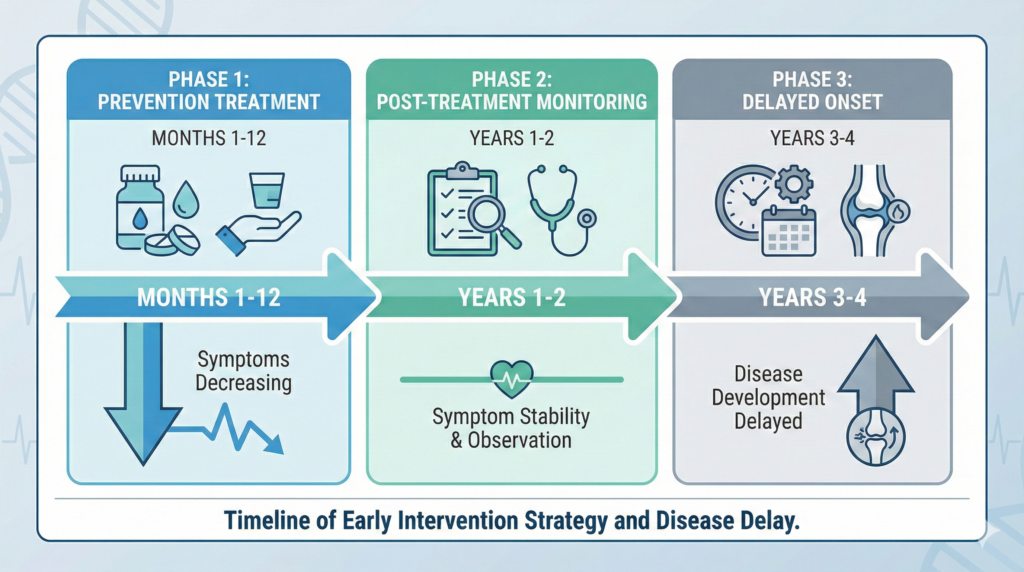
The APIPPRA trial, published this month by researchers at King’s College London, followed 213 high-risk individuals for up to eight years—making it the longest study of its kind. The results? Those who received just one year of prevention treatment with abatacept went on to delay rheumatoid arthritis by four full years after treatment stopped.
For the 17.9 million people worldwide living with RA, this represents hope. For those at high risk, it’s a game-changer.
But here’s what most people don’t realize: rheumatoid arthritis doesn’t just cause joint pain. It eats away at the bone underneath your joint lining, eventually causing permanent deformity and fusion. It can damage your heart, lungs, and eyes. And once bone erosion begins, there’s no reversing it.
That’s why early intervention matters. That’s why this breakthrough could save millions from a lifetime of pain.
The Study That’s Rewriting Medical Textbooks
The numbers tell a story that’s hard to ignore.
Researchers recruited 213 people from the UK and Netherlands—all considered high-risk for developing rheumatoid arthritis. These weren’t people with full-blown RA yet. They were individuals with suspicious joint symptoms, family history, and most importantly, specific antibodies in their blood that signal immune system dysfunction.
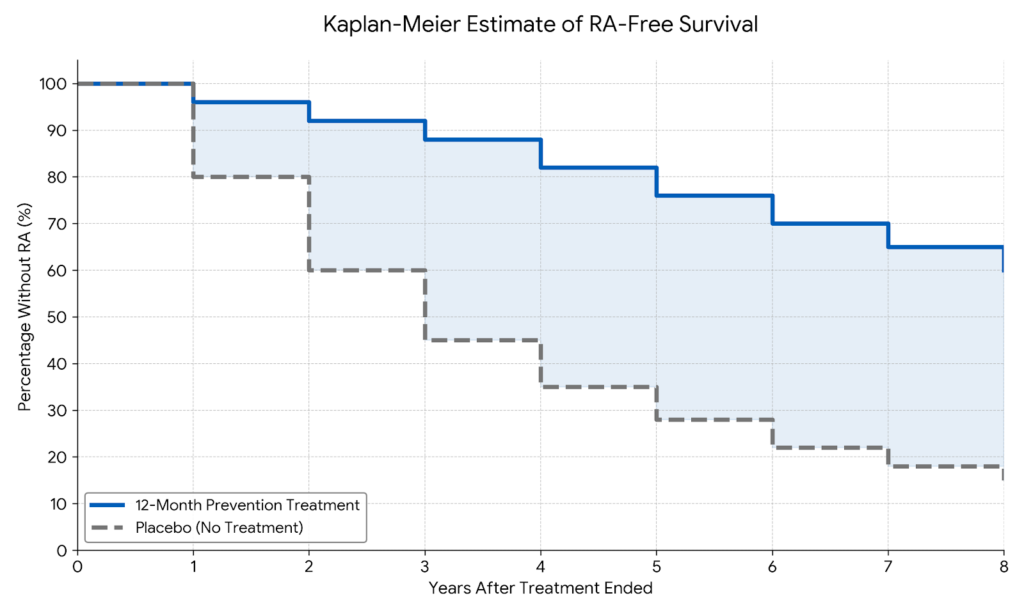
Half received abatacept, a medication that modulates the immune system. Half received a placebo. Both groups were treated for exactly 12 months, then researchers followed them for years.
The results stunned the medical community.
Those who received abatacept took significantly longer to develop rheumatoid arthritis compared to the placebo group—with disease onset delayed by up to four years beyond the treatment period. Even more remarkable, the people with the highest antibody levels—those at greatest risk—benefited the most.
Dr. Andrew Cope, professor of rheumatology at King’s College London and lead researcher, put it bluntly: “Intervening early in people at high risk can have lasting benefits. We have shown that this approach is safe and can prevent disease while patients are on treatment as well as substantially relieve symptoms. Importantly, it can delay the onset of RA for several years, even after treatment has stopped.”
Think about what four years means. Four years of normal hand function. Four years without daily medication. Four years to advance your career, play with your children, button your own shirt without pain. Four years before your immune system begins its assault on your joints.
Dr. Orrin Troum, a board-certified rheumatologist not involved in the study, called it “definitive proof for delaying the onset of autoimmune diseases.” He emphasized that finding ways to delay RA is critical because it addresses the lifelong burden of a chronic, incurable disease before irreversible damage occurs.
What Exactly Is Rheumatoid Arthritis?
Before we go further, let’s understand what we’re dealing with.
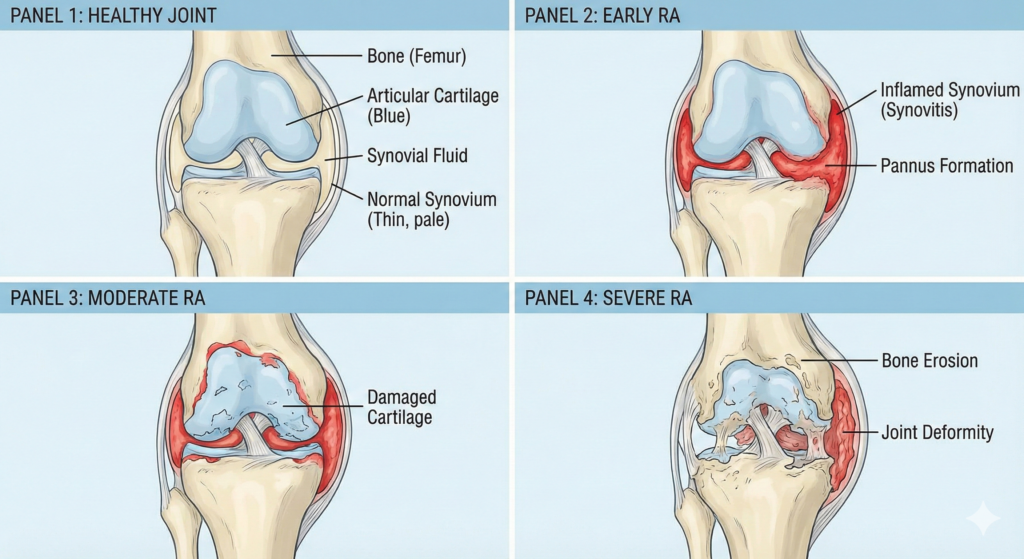
Rheumatoid arthritis is a chronic autoimmune disease where your immune system attacks the tissue lining your joints by mistake. Imagine your body’s defense system—designed to protect you from viruses and bacteria—suddenly turning its weapons on your own tissue.
The target? A thin membrane called the synovium that lines your joints and produces lubricating fluid. When your immune system sends antibodies to attack this lining, inflammation occurs. The synovium swells and thickens. Fluid builds up. Your joints become painful, warm, and stiff.
But that’s just the beginning.
Over time, the chronic inflammation damages the cartilage between your bones—the cushioning that acts as your joints’ shock absorber. As cartilage deteriorates, the space between bones narrows. Eventually, the disease eats away at the bone itself, causing joints to deform, shift out of place, and in severe cases, fuse together permanently.
This isn’t wear-and-tear arthritis from aging. That’s osteoarthritis—a completely different disease. Rheumatoid arthritis is an immune system malfunction that can strike people in their thirties, forties, and fifties. Your most productive years.
And it doesn’t stop at joints.
RA can inflame and scar your lung tissue, making it progressively harder to breathe. It can damage the sac around your heart and harden your arteries, raising your risk of heart disease. It can cause dry eyes so severe you can barely see. It can form hard nodules of tissue—not just under your skin, but on your heart and lungs.
Without treatment, rheumatoid arthritis is a progressive disease that steals your ability to do the simplest daily tasks. With treatment, many people achieve remission. But prevention? Until now, that was off the table.
The Silent Alarm Most People Ignore
Cleveland Clinic doctors describe early RA symptoms as “like an alarm clock set to vibrate”—subtle enough to dismiss, but important enough to catch.
So what does that vibrating alarm sound like?
Morning stiffness that won’t quit. We’re not talking about the few minutes of stiffness you feel when getting out of bed. This is stiffness lasting 45 minutes, an hour, sometimes longer. Your hands feel like they’re wearing invisible gloves. Your feet feel like they’re encased in concrete. Movement helps, but it takes time.
Symmetrical joint pain. Both hands hurt. Both feet ache. The same knuckles on your left and right fingers throb. This pattern—affecting the same joints on both sides of your body—is RA’s signature. Osteoarthritis typically affects one side more than the other.
Small joints affected first. Your fingers, toes, and wrists bear the brunt of early RA. Later, it can spread to knees, elbows, ankles, shoulders, and hips. But those small joints? They’re usually where the attack begins.
Fatigue that feels like walking through concrete. Not just tiredness. This is profound exhaustion that doesn’t improve with rest. Some days, getting out of bed feels impossible. Your body is fighting itself, and that internal war drains your energy.
Other symptoms people overlook. Low-grade fever. Loss of appetite. Unintended weight loss. Tender, warm joints that hurt when you apply pressure.
Here’s the critical part: these symptoms come and go in cycles called flares and remissions. You might have weeks of pain followed by weeks of feeling normal. This pattern tricks people into thinking it’s not serious. They tell themselves it’s stress, or aging, or too much time on the computer.
Meanwhile, their immune system is ramping up its attack. Inflammation is damaging cartilage. Bone erosion is beginning—silently, invisibly.
The difference between catching RA in its early stages and waiting until joints deform? That’s the difference between managing a condition and living with permanent disability.

Who’s Actually at Risk?
Not everyone faces the same odds of developing rheumatoid arthritis. Certain factors dramatically increase your risk.
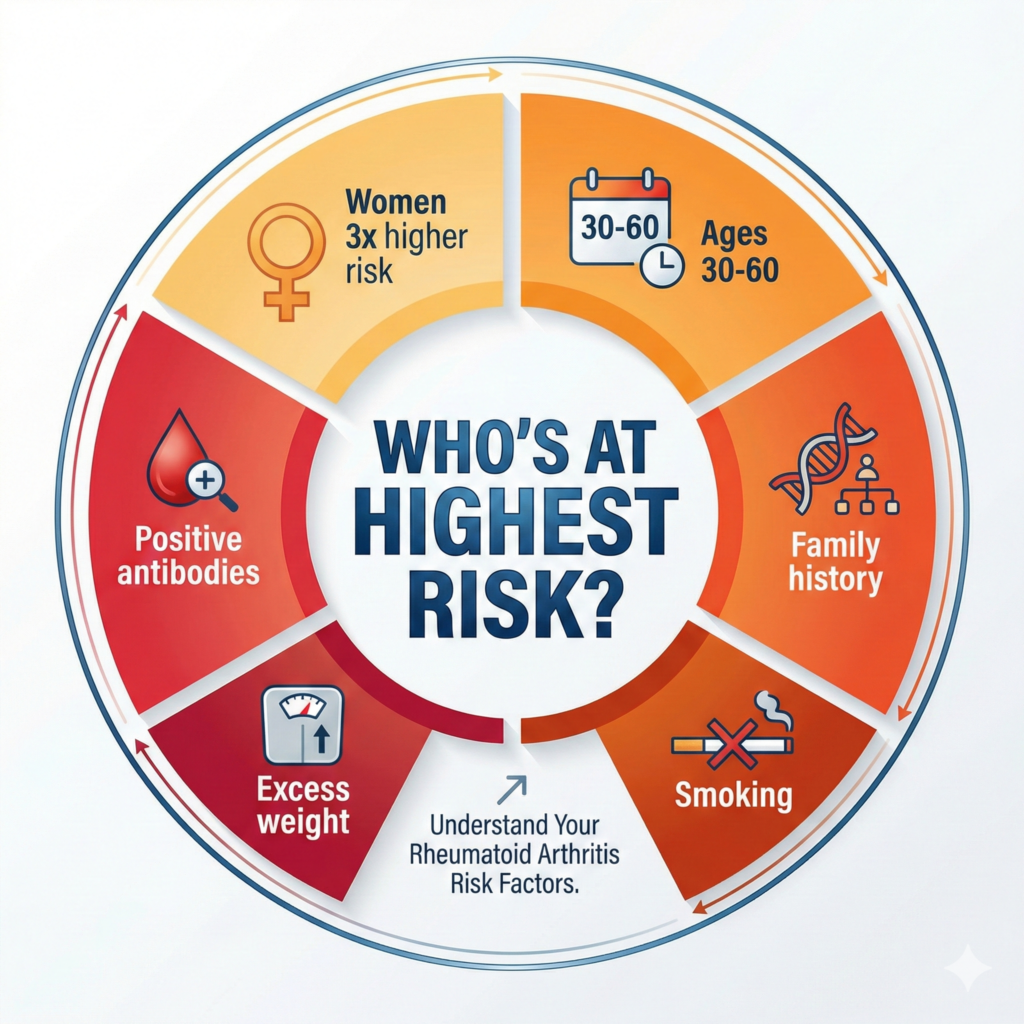
Gender matters—a lot. Women are three times more likely than men to develop RA. Seventy percent of people living with rheumatoid arthritis are women. Researchers believe hormones play a role, though the exact mechanism remains unclear.
Age narrows the window. Most people develop RA between ages 30 and 60—peak working years, peak parenting years, peak life years. When it affects people between 16 and 40, doctors call it young-onset RA or YORA. After age 60, it’s later-onset RA or LORA.
Family history can’t be ignored. If your parent or sibling has rheumatoid arthritis, your risk shoots up. The disease runs in families because of specific genetic variations, particularly in genes called HLA genes. These genes help your immune system distinguish your body’s proteins from invader proteins. When they malfunction, your immune system gets confused.
Smoking is the biggest modifiable risk factor. Cigarette smoking over time significantly raises your risk of developing RA. Even worse, smoking makes the disease more severe in people who already have it. If you smoke and have other risk factors, quitting isn’t optional—it’s essential.
Other risk factors stack up. Serious gum infections raise RA risk. Being overweight increases susceptibility. Exposure to certain environmental pollutants and irritants may trigger the disease in genetically vulnerable people. Lower socioeconomic status correlates with higher rates, possibly due to environmental factors and healthcare access.
But here’s what makes the prevention study so powerful: it identified people at highest risk using blood tests that detect specific antibodies. Those antibodies signal that your immune system is already going haywire—you just don’t have full symptoms yet.
The people with the most antibodies? They got the most benefit from prevention treatment. That’s precision medicine in action—using biomarkers to identify exactly who needs intervention.
The Prevention Revolution: What It Actually Means
Let’s break down what this prevention treatment does—and doesn’t—do.
What happened in the study: High-risk individuals received abatacept infusions or injections for 12 months. Abatacept works by blocking a signal that activates T-cells, a type of immune cell involved in the autoimmune attack. By calming this immune response early, before full disease develops, researchers hoped to change the disease trajectory.
It worked.
During the treatment year, participants experienced reduced joint pain and fatigue—even though they hadn’t been diagnosed with full RA yet. Their bodies were already fighting inflammation, and the medication was winning.
After treatment stopped, researchers continued monitoring. This is where the magic happened. The benefits didn’t immediately disappear. Instead, people who received abatacept took significantly longer to develop rheumatoid arthritis compared to those who got placebo.
Up to four years longer.
Think about the implications. Four extra years means four more years of preserved cartilage. Four years before bone erosion begins. Four years of maintaining your quality of life, your career, your ability to do the things you love.
Dr. Troum emphasized what this means in practical terms: “Delaying RA onset by four years preserves joint function and mobility, improves quality of life, prevents systemic complications, sustains employment and productivity, reduces long-term healthcare costs, and optimizes personalized medicine.”
But there’s a catch. Once treatment stopped, symptom levels gradually returned to similar levels as the placebo group. This suggests that the immune modulation effect is temporary without continued treatment. The disease was delayed, not cured.
This raises important questions. Should high-risk people receive ongoing treatment to maintain prevention? How long is optimal? What are the long-term effects of extended immune suppression in otherwise healthy people?
These questions are exactly what researchers plan to investigate next.
The Ethical Debate Doctors Are Having Right Now
Here’s where it gets complicated.
Not everyone with RA antibodies develops the disease. Studies show that a significant percentage of people with suspicious symptoms and positive antibody tests never progress to full rheumatoid arthritis.
So should we treat them anyway?
Dr. Arthur Kavanaugh, a rheumatologist at UC San Diego not involved in the study, raised this exact concern: “The challenge is that a number of patients with clinically suspect arthralgia do not go on to develop RA, so this raises ethical questions about treating patients to prevent a disease that they may never develop.”
It’s a valid point. We’re talking about giving immunosuppressant medications to people who are technically healthy. These medications have side effects. They increase infection risk. They require regular monitoring.
On the other hand, for those who do develop RA, the four-year delay could be life-changing.
The key is precision. The APIPPRA study showed that people with the highest antibody levels—the truly high-risk group—benefited most. As screening tools improve, doctors will get better at identifying who really needs prevention versus who can safely be monitored.
This is personalized medicine at work. Not everyone gets the same treatment. Instead, blood tests, genetic markers, family history, and symptom patterns combine to create a risk profile. Those at highest risk get the most aggressive intervention.
The medical community is wrestling with these questions in real time. Guidelines are being written. Risk calculators are being developed. Screening infrastructure is being built.
What’s clear is this: for the first time, we have proof that autoimmune disease can be delayed before it starts. Now we need to figure out how to do it responsibly.
What You Can Do Right Now
Knowledge without action is just interesting trivia. Here’s what you should actually do with this information.
If you have RA family history, get tested. Ask your doctor about antibody screening. Tests like rheumatoid factor, anti-CCP antibodies, ESR, and CRP can detect immune system problems before symptoms become severe. If antibodies are high and you have early symptoms, you may qualify for prevention protocols.
If you have symptoms, don’t wait. Morning stiffness lasting 45 minutes or more? Joint pain affecting the same areas on both sides of your body? Persistent fatigue and low-grade fever? See a rheumatologist, not just your general practitioner. Rheumatologists specialize in these conditions and can order comprehensive testing and imaging.
Stop smoking immediately. This isn’t negotiable. Smoking is the single biggest modifiable risk factor for RA. It raises your risk and makes the disease worse if you develop it. Every cigarette is another trigger for your immune system.
Track your symptoms carefully. Use your phone to note when stiffness occurs, how long it lasts, which joints hurt, and whether symptoms are symmetrical. This information helps doctors make faster, more accurate diagnoses.
Ask about family history. Talk to your parents and siblings. Do they have RA or other autoimmune diseases? This information helps your doctor assess your risk level.
Maintain a healthy weight and eat anti-inflammatory foods. While diet can’t cure RA, it can help manage inflammation. Focus on lean proteins, omega-3 rich fish, colorful vegetables, fruits, and whole grains. Minimize processed foods, refined sugars, and saturated fats.
Move your body regularly. Contrary to myth, exercise doesn’t make RA worse—it makes it better. Low-impact activities like swimming, yoga, walking, and tai chi reduce inflammation and keep joints functional.
Join support communities. Nearly half of RA patients find valuable support and information through online communities. You’re not alone in this. Learning from others’ experiences can help you navigate diagnosis and treatment.
Most importantly, trust that subtle alarm. Your body is trying to tell you something. The stiffness, the fatigue, the symmetrical pain—these aren’t just aging or stress. They might be your immune system’s first attacks on your joints.
Catch it early, and you have options. Wait too long, and bone damage becomes permanent.
Beyond Joints: The Full Body Impact
Here’s what many people don’t know: rheumatoid arthritis is a systemic disease. Yes, it attacks your joints. But the same inflammatory chemicals circulating through your bloodstream can damage organs throughout your body.
Your heart is at risk. RA significantly increases your chances of developing heart disease. The chronic inflammation can harden and block arteries. It can inflame the pericardium, the protective sac around your heart. Many RA patients don’t realize that cardiovascular complications are one of the leading causes of death in people with the disease.
Your lungs can scar. RA-related inflammation can affect lung tissue, causing scarring that progressively worsens over time. This leads to shortness of breath that gets worse with activity. Some people develop nodules on their lungs. Others experience inflammation of the lining around the lungs.
Your eyes get dry and inflamed. Many RA patients develop secondary Sjogren’s syndrome, a condition that dramatically reduces moisture in eyes and mouth. Severe dry eye isn’t just uncomfortable—it can affect your vision and quality of life.
Rheumatoid nodules form in unexpected places. These firm bumps of tissue usually appear on elbows, but they can develop anywhere—including on your heart, lungs, and other internal organs.
Your cancer risk increases. People with RA have higher rates of lymphoma and possibly other cancers. Whether this is due to the disease itself or the immunosuppressant medications used to treat it remains unclear.
Infections become more frequent. RA impairs immune function, and the medications used to treat it suppress immunity further. This creates vulnerability to infections ranging from common colds to serious bacterial infections.
Osteoporosis weakens your bones. Both RA itself and corticosteroid medications used to manage flares increase your risk of bone loss. This makes fractures more likely.
Carpal tunnel syndrome compresses nerves. When RA affects your wrists, swelling can press on the median nerve, causing numbness, tingling, and pain in your hand and fingers.
This is why delaying disease onset matters so much. Every year you delay is a year your heart, lungs, eyes, and bones remain undamaged. Every year without systemic inflammation is a year of better overall health.
The Treatment Landscape Today
If you do develop rheumatoid arthritis, here’s the good news: treatment has improved dramatically over the past two decades.
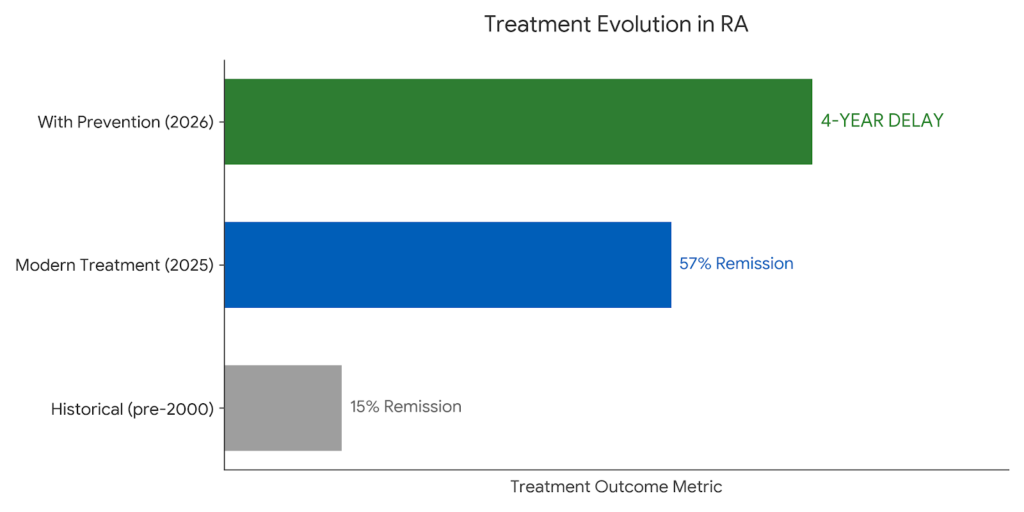
Gone are the days when RA meant inevitable disability. Modern medications, used aggressively and early, can put the disease into remission in over half of patients.
Disease-modifying anti-rheumatic drugs or DMARDs form the foundation of treatment. Methotrexate is the most common. These medications don’t just mask symptoms—they slow or stop the disease process itself. Starting DMARDs early prevents joint damage.
Biologic medications target specific parts of the immune response. Unlike older drugs that broadly suppress immunity, biologics precisely block the inflammatory chemicals driving joint destruction. TNF blockers, IL-6 inhibitors, and other biologics have revolutionized outcomes.
JAK inhibitors represent the newest class of medications. These oral drugs block enzymes involved in the inflammatory process. They offer another option for people who don’t respond to traditional DMARDs or biologics.
Corticosteroids remain valuable for controlling flares—sudden worsening of symptoms. However, doctors use them sparingly due to long-term side effects like bone loss and increased infection risk.
The treat-to-target approach has become standard care. Instead of just managing symptoms, rheumatologists aim for remission or very low disease activity. This aggressive strategy, with regular monitoring and medication adjustments, achieves sustained remission in 57 to 65 percent of patients.
Surgery is reserved for severe cases. When joints are too damaged or painful to respond to medication, options include joint replacement, tendon repair, or synovectomy—surgical removal of inflamed joint lining.
The goal isn’t just reducing pain. It’s preserving function, preventing deformity, and maintaining quality of life. With early diagnosis and proper treatment, most people with RA can continue working, staying active, and doing the things they love.
But prevention beats even the best treatment. Why live with a manageable disease when you could delay it by years?
Hope on the Horizon
The prevention study is just the beginning.
Researchers are already working on the next wave of innovations that could make RA even more preventable and treatable.
Gene therapy targeting TRAF1 shows promise in early studies. This approach could potentially eliminate the inflammatory response at its genetic source, offering hope for a future cure rather than just management.
Tolerogenic dendritic cell therapy aims to “reset” the immune system in autoimmune diseases. By retraining immune cells to tolerate the body’s own tissues, this approach could stop the autoimmune attack permanently.
Refined risk stratification tools will help doctors better identify who truly needs prevention treatment. As we understand more about genetic markers, antibody patterns, and environmental triggers, prediction will become more precise.
Optimal duration studies are underway to determine how long prevention treatment should last. Should it be 12 months? 24 months? Should it be continuous for high-risk individuals? These questions will shape future protocols.
Non-drug interventions are being explored. Can specific diets prevent or delay RA in high-risk people? What about stress reduction techniques? Exercise protocols? The answers could provide additional tools beyond medication.
Digital and decentralized trial models will make research more accessible. Instead of requiring participants to travel to major medical centers, remote monitoring and telemedicine can include people from diverse locations and backgrounds.
Screening infrastructure improvements will make prevention accessible to more people. As awareness grows and testing becomes routine for high-risk individuals, more people will get the early intervention they need.
The field is moving fast. What seemed impossible five years ago—preventing an autoimmune disease before it starts—is now proven science. What seems impossible today might be routine a decade from now.
The Bottom Line
Rheumatoid arthritis affects 17.9 million people worldwide. For decades, there was no way to prevent it, especially if it ran in your family. The best doctors could do was diagnose early and treat aggressively.
That just changed.
The APIPPRA trial proved that treating high-risk individuals for just 12 months can delay disease onset by up to four years. Those with the highest risk—detected through simple blood tests—benefit the most.
This is the definition of a medical breakthrough. A paradigm shift from reactive treatment to proactive prevention. A chance to preserve joints, protect organs, and maintain quality of life before irreversible damage occurs.
But breakthroughs only help people who know about them.
If you have RA family history, get tested. If you have morning stiffness lasting 45 minutes or more, see a rheumatologist. If you’re smoking, quit. If you notice symmetrical joint pain, don’t dismiss it as stress or aging.
That subtle alarm your body is sounding? Listen to it.
Early intervention is the difference between managing a chronic disease and delaying it by years. Between preserved function and permanent deformity. Between independence and disability.
The science is clear. The evidence is strong. The opportunity is here.
What you do with this information is up to you.
Medical Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider regarding diagnosis, treatment, and medical decisions. The information presented here is based on published research and expert opinions current as of January 2026.
Continue reading to learn about treatment options...

No Comments Yet
Be the first to share your thoughts!